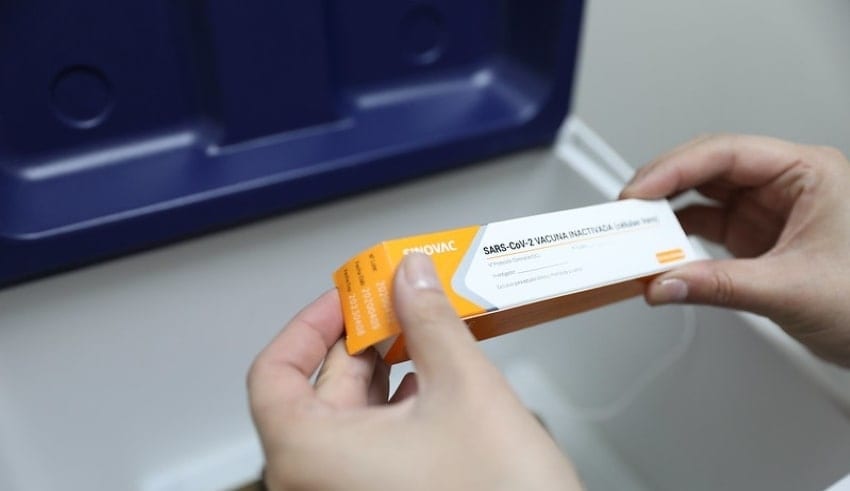
In the wake of getting crisis use approval, China’s Sinovac poke needs to go through specialized appraisal and assessment before inoculation, as per the Healthcare Professionals Alliance Against COVID-19.
Dans shared there is no compelling reason to look for endorsement of the Inter-Agency Task Force for the specialized survey in light of the fact that the HTAC is ordered by law to assess. He clarified that the FDA evaluates principally the wellbeing and adequacy of immunizations while the HTAC survey goes past wellbeing concerns.
In the wake of getting crisis use approval (EUA), China’s Sinovac poke needs to go through specialized appraisal and assessment before inoculation, as per the Healthcare Professionals Alliance Against COVID-19 (HPAAC).
Currently, they are in talks with Department of Health, Health Technology Assessment Council and Food and Drug Administration and I think there is general arrangement that regardless of whether it’s given, it ought to go through HTAC assessment. It’s the subsequent advance.
In the interim, a chief creator of the recently passed Emergency Vaccine Procurement Act disclosed that antibodies to be bought under the measure can’t be utilized for business purposes.
At the present time there is an unusual inventory circumstance, so if private substances will buy, they can utilize it just for their workers for the time being and not sell it.
Consequently, Cua called the attention to that unifying obtainment of immunizations through the public government is the most effective method of securing the pokes. Also, albeit the measure permits acquisition and acquisition of antibodies to swear off open offering during a pandemic, these are as yet dependent upon a review.
Along with this, Senate Minority Leader Franklin Drilon shared the public authority can’t give outright and cover insusceptibility to antibody producers since it is illegal and in spite of public strategy.
Under the COVID-19 Vaccination Program Act Congress passed last Feb. 22nd COVID-19 antibody makers are resistant from suits for claims emerging out of the organization of the COVID-19 immunization, however not for headstrong offense or gross carelessness.
Morever, Drilon clarified that any vaccinee can record claims for harms, in view of the antibody producers liabilities emerging from stiff-necked wrongdoing and gross carelessness.
























