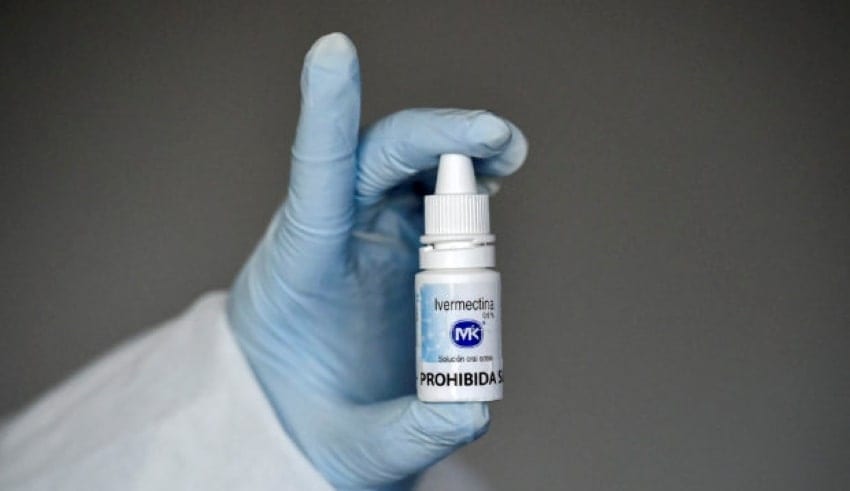
The Department of Health (DOH) has effectively acquired outcomes from the specialists’ assessment of ivermectin for COVID-19 treatment, however the outcomes presently can’t seem to be finished, Health Undersecretary Maria Rosario Vergeire shared it to the public.
She highlighted that their specialists have been assessing this ivermectin for these previous weeks and we have as of now results from this assessment. We simply need to conclude the report and afterward, we will advise the general society.
Related Posts
Vergeire highlighted that the Philippine COVID-19 Living CPG Reviewers’ assessment of ivermectin is practically finished. Along with this, She said that the outcomes might be out in the coming days or weeks. She at that point encouraged general society to sit tight for ivermectin to be enrolled for COVID-19 avoidance and treatment in the country prior to utilizing it against the respiratory illness.
This is exactly the thing they are saying, They should simply sit tight for the inform regarding our specialists. It won’t be long. How about we simply trust that this medication will be enlisted here in the country so government can likewise have the option to screen and have the option to be responsible if at any point someone will take that drug? That was te continuous questions arised.
Vergeire focused on that the public authority can’t ensure the security of ivermectin as it is as yet unregistered for human use in the country. She added that those utilizing the item may likewise have a misguided feeling of security against COVID-19. They base everything on science as they can’t simply agree to all that in light of the fact that different nations are utilizing it, since the entirety of their residents are now utilizing it then they simply take the path of least resistance and give them the go sign. For this reason, The DOH and FDA (Food and Drug Administration) has that order of securing the populace, general wellbeing, and we have that command of guaranteeing that these medications that you take will be protected and of value. The FDA prior conceded an empathetic unique license for a medical clinic’s utilization of ivermectin for COVID-19 patients.