Thailand’s administration experienced harsh criticism in Parliament over its Covid immunization strategy, with resistance administrators blaming experts for being too delayed to even think about beginning vaccinations and depending overwhelmingly on a solitary antibody maker.
For this reason, Thailand has requested 2,000,000 dosages from Sinovac Biotech for its initial antibody turn out and has gotten 61 million portions from AstraZeneca for its mass vaccination plan, delivering those locally from June through regal possessed firm, Siam
Mr Wiroj shared that territorial neighbors Myanmar, Indonesia and Cambodia had just begun inoculations. Thailand’s methodology has stood out forcefully from those of most South-east Asian nations, which have tried to get conveyances before, from different suppliers. The Philippines is in talks with seven antibody producers, while Malaysia has supply concurrences with fi
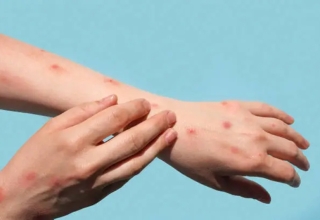
Thailand has had far less contaminations, notwithstanding, with 24,961 cases generally speaking and 82 passings. A representative for the public authority on Sunday safeguarded its choice not to join the global antibody sharing office Covax, saying it might have brought about more exorbitant costs and dubious conveyance dates.
Along with this, the Health Minister Anutin Charnvirakul dismissed the analysis in Parliament and said the public authority’s arrangement ensured adequate inventory. He said the AstraZeneca understanding would pay off, with an innovation move that would help Thailand become an antibody creation center.
Pundits have also scrutinized the reasonableness of Siam Bioscience as the nearby maker as it still can’t seem to make antibodies. The director of the National Vaccine Institute, Dr Nakorn Premsri, showed reporters a letter that he said was from AstraZeneca, saying that over 60 prospective global partners had been checked and Siam Bioscience was among the 25 chosen by it.
Previously, AstraZeneca highlighted that Siam Bioscience had undergone a virtual five-day audit and all the lots of vaccines it manufactures will undergo safety checking which is something to observe for the next days.